Whisky Flavor

Distilling
As I mentioned in my last article, whisky draws its flavor from three sources – ingredients, distillation process, and aging. Nothing more, nothing less. Today, let’s look at the effects of distilling on the final product. So how exactly does the distilling affect the taste of your favorite dram? Well, it’s sort of an art AND a science. Some tradition and some modern technology, all mixed up with a dash of chemistry and a hint of physics.
Lets start with Physics 101 – Latent Heat of Vaporization. In a nutshell, this is how water turns into steam (and how alcohol turns into booze-steam).
If we mix up a whisky mash, we end up, in a few days, thanks to the magic of yeast, with a mix of ethanol (alcohol) and water. We use distillation to separate one from the other. It’s pretty easy. The boiling point of alcohol is 173F (78c). The boiling point of water is 212F (100C). So, when we add some heat to the still, the physics part kicks in (Second Law of Thermodynamics, for anyone who was wondering). No matter how much we heat the still, all the alcohol will evaporate before any of the water. Once the mixture hits 173F, the alcohol starts to boil. This boiling alcohol absorbs all of the energy from the heat. So the temperature won’t go up until all of the alcohol is evaporated. Well, that’s what mostly happens. We’ll talk more about the details in the chemistry lesson. Anyway, as the alcohol boils, it turns into a sort of booze-steam that rises up to the top of the still. It then cools and condenses as it enters the cooling pipe that’s attached the the top. The cooled booze-steam then condenses back into alcohol and runs down the pipe into a collection chamber. And that liquid, ladies and gentlemen, is the freshly made whisky-to-be.
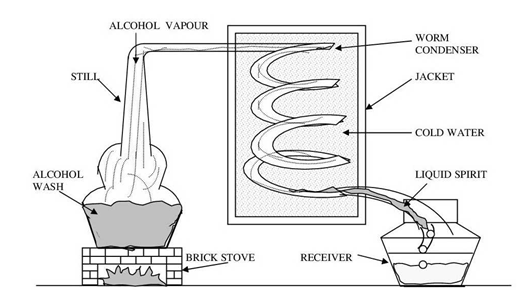
Pretty simple, right? Not so fast. We need to have a quick chemistry lesson to account for the transfer of flavor.
The physics part should mean that only pure alcohol comes out of the still. And pure alcohol wouldn’t have any of the flavor from the grain or the smoke, or the special water supplies that we talked about in my last article. So how does the flavor get through distillation? Simply put, it hitches a ride. Before distillation, we have a mixture of water and yeast-created-alcohol. Mostly. But there are also lots a other things in there, like fats and oil droplets from the grain, phenol molecules from the smoke, esters from the fermentation process, and a few other compounds that carry flavor and affect taste.
These various molecules start to get busy as the still starts heats up. Some will break down into smaller molecules, others form chemical bonds and stick to the alcohol molecules, and some just get stuck between other molecules and catch a ride in the steam. And, depending on the shape of the still and the skill of the person running the still, many of them make it all the way into the cooling tube and into the collector.
A rule of thumb – the taller the still, the lighter the taste of the whisky. That’s because the heavy molecules, like the fats and oils, aren’t light enough to survive the trip to the top. And some of the smaller, flavor-rich compounds like to bond to the fats and oils rather than to the alcohol molecules, so they don’t make it through the process either. That’s why the heavy tasting, smoky whiskies tend to be made in short stills, where all these delicious flavors are more likely to survive the process.
So, what have we learned from all this? In a nutshell – distillation has an impact of the flavor of our favorite whisky, thanks in part to physics, chemistry, the shape of the still, and the process. Along with the skill of the still operator in pushing all of the right buttons at the right time. Trust me – our favorite whisky companies have been tweaking the process for a long time, to get their trademark taste just right.
Next, we’ll take a peek at the aging process, where most of our whisky flavor comes from. Until then, cheers, and enjoy!
Thanks, I learned more from this than in all my high school and university courses in chemistry…. But to be fair I didn’t remember much even then.
LikeLike
Love the term “booze-steam”
LikeLike